Excipients in Peptide Science: Roles, Functions, and Research Insights
Understanding Excipients
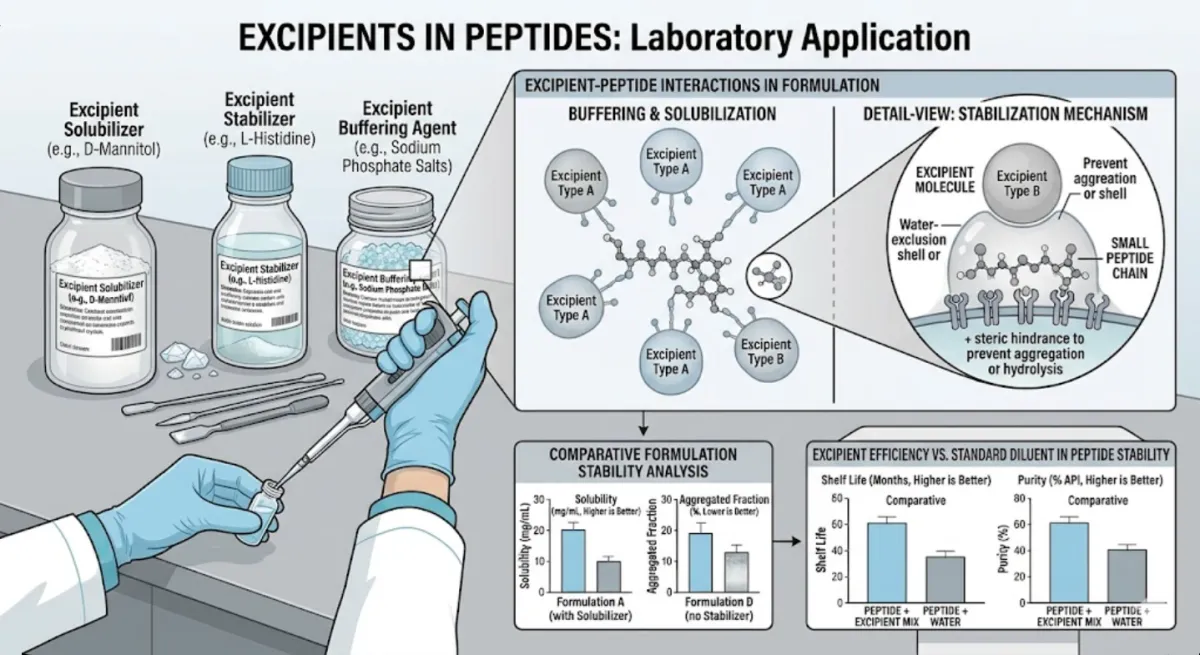
Excipients are the “supporting cast” in peptide formulations. While peptides are the active molecules, excipients ensure stability, usability, and reproducibility. They can act as carriers, stabilizers, or buffers, helping peptides maintain their structure and function during storage, transport, and research applications. At HELIX Source, excipients are treated as essential components of reliable peptide science.
How Excipients Work
Excipients serve multiple purposes depending on the formulation. Some prevent peptide degradation by controlling pH or moisture. Others act as bulking agents, making peptides easier to handle in lyophilized form. Certain excipients improve solubility, ensuring peptides can be reconstituted consistently in laboratory settings. Together, they create a controlled environment that protects delicate molecules.
Research Findings
Studies have shown that excipients can significantly extend peptide shelf life and improve reproducibility across experiments. Common excipients include mannitol, trehalose, and glycine, each chosen for specific stabilizing properties. Research also highlights the importance of excipient compatibility, as mismatched additives can interfere with peptide activity. Transparency in excipient use is therefore critical for compliance and reproducibility.
Why It Matters
Excipients may not be the headline molecules, but they are vital for ensuring peptides perform as expected in research. By documenting excipient use and integrating QR‑coded Certificates of Analysis, HELIX Source provides researchers with full visibility into formulation details, reinforcing trust and scientific credibility.
Looking Ahead
As peptide science advances, excipient research continues to evolve. New stabilizers and carriers are being tested to improve efficiency, reduce variability, and align with sustainability goals. HELIX Source remains committed to adopting these innovations to support cutting‑edge research.
References
Wang, W. (2000). Lyophilization and development of solid protein pharmaceuticals. International Journal of Pharmaceutics.
Carpenter, J.F., et al. (2002). Rational design of stable lyophilized protein formulations: theory and practice. Pharmaceutical Biotechnology.
Pikal, M.J. (1990). Role of excipients in peptide and protein stability. Biotechnology Progress.
For Research Use Only – Not for Human or Animal Use. The contents of this article is for educational and informational purposes only.
